Enrollment & Retention
Hit your critical trial milestones on time.
Find the right participants who will take your study over the finish line.

Connect the right participants to the right studies at the right time.
According to industry research, 80% of all clinical trials fail to reach their enrollment targets. With trial activity increasing but performance dropping, the ability to connect participants to the right studies is a major challenge – and one that puts the success of studies at risk for sponsors, CROs, and sites.
Take charge of your study’s outcome. WCG’s end-to-end enrollment and retention solutions are the best way to find (and keep) your trial’s perfect matches. Meet critical milestones, avoid costly shortfalls, and get the make-or-break trial data you need.
Explore our Enrollment & Retention Solutions
Participant Identification
Participant Recruitment & Retention
Diversity, Equity, & Inclusion
Study Advertising
Enrollment & Retention Results:
56% Faster Enrollment
WCG accelerated enrollment for complex vaccine study
500+ Referred Participants
WCG referred participants in just 30 days for healthy volunteer study
34% Increased Enrollment
WCG increased enrollment for major depressive disorder study
Meet the technology that powers our Enrollment & Retention solutions
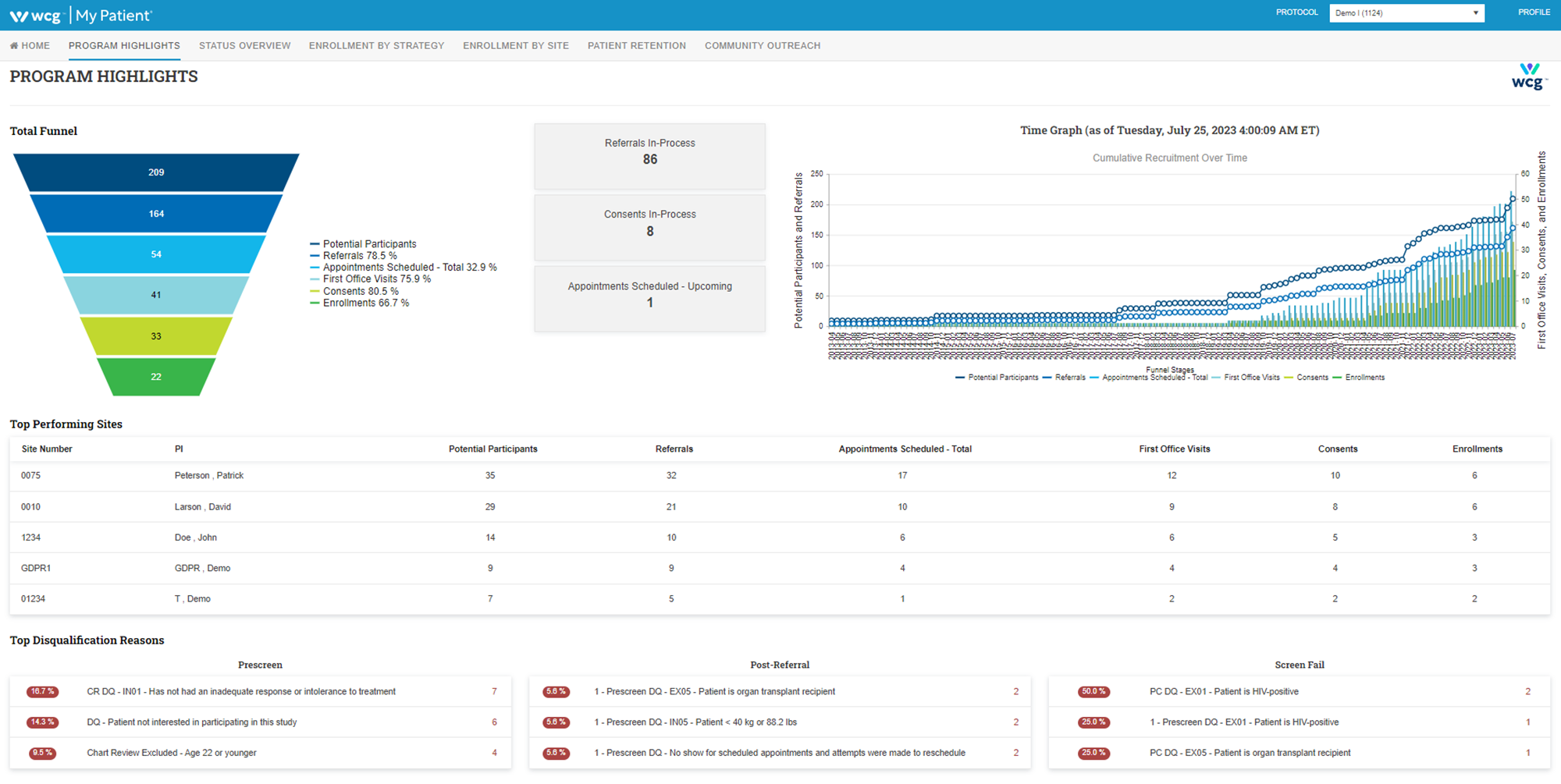
My Patient®
Monitor each stage of the participant journey in real-time and at the most granular level possible. Access advanced metrics and actionable insights – anywhere, from any device, using a simple, intuitive user interface.
Designed to support the participant journey, My Patient is the central hub of any participant recruitment campaign. With 24-7 real-time data visible through a secure web-accessed study management portal, sites, sponsors, and CROs now have more visibility and control over the recruitment process.
Customer Stories
“…You have exceeded our expectations and have delivered remarkably during these past six months. When you got involved we were struggling more than I’ve ever experienced in any trial, and the manner in which you took ownership of this challenge was impressive.”
Executive Director, Top 5 Pharma Company
What is the ultimate goal of our clinical leadership? On-time enrollment. By collaborating with WCG, I truly believe that we are breaking down the barriers to efficiency, and in doing so, the barriers to on-time enrollment.”
Senior Director, Global Development Operations, Mid-Size Pharmaceutical Company
Unlike many other participant recruitment companies, WCG truly delivers on their projections… they are adept at placing media in the most targeted and efficient manner to deliver high-quality referrals to the site.”
Recruitment Lead, Top 10 Pharma Company
Connect the right participants to the right studies at the right time.
Our team of experts is ready to discuss your studies and how we can quickly meet patient enrollment milestones. Complete the form to schedule a consultation with WCG.